|
|
Why did the bee cross the road? In order to answer this question first consider the road. On the first day working with the Echinacea Project, we (Zeke, John, and Brigid) visited the Around Landfill site. The aforementioned road passes through this Around Landfill site, which coincidentally, is located around a landfill. On either side of the road, C4 warm season plants and legumes grow. Alfalfa and Yellow Sweet Clover pepper the roadside, breaking up the green with flashes of purple and yellow. Small Echinacea buds hint at flowers to come. Corn dominates the landscape farther from the road. Cattle once grazed on one side of this road, as evidenced by a barbed wire fence.
There wasn’t always a road to cross. Before the road, a sea of uninterrupted prairie swayed as far as the eye could see. At this time, before the road broke the continuity of the prairie, the bee had no need to cross the road. Now, grasses, flora and legumes must straddle the road. In order to bridge this divide and visit flowers, the bee must now cross the road.
But can bees cross the road? Consider this question from the perspective of a bee. A bee’s umwelt is quite different from that of humans; bees have a short field of vision as well as the ability to see the UV spectrum of light.
We all sat on the roadside at this site, wondering if bees were indeed able to cross the road. Then, just when we asked this question aloud, a butterfly serendipitously flew across the road, as if in answer. If a butterfly can cross, we supposed that a bee can as well.
But will the bees be able to cross the road in the future? Given rapid expansion of cities, it is likely that the road will wide, creating a world for cars and not bees. Getting to ‘the other side’ could prove a much more daunting task in the future. 
Now that my time at the garden is coming to an end, I wanted to include a summary of the projects that I’ve been working on. These three projects include organizing solitary bees that have been collected from yellow pan traps in Minnesota over the past summer, identifying pollen on Echinacea styles and recording the behavior of solitary bees inside emergence traps.
Last summer, several yellow pan traps were placed on the sides of roads in Minnesota in hopes of collecting solitary bees. Once they were collected, each solitary bee was pinned and tagged with a label that included the date, trap number, location, and an ID code. Before logging any of the information into the Roadside Pollinator spreadsheet (this keeps track all of the solitary bees that were collected over the summer in the pan traps) I grouped the specimen together by  taxon. I started grouping more recognizable groups together like Agapostemon virescens before looking at more difficult specimen. Once all grouped together, I would enter the information listed on the label into the spreadsheet. I started with the specimen at the top left corner and worked my way to the bottom right corner of the box. I did it this way to make it easier for anybody to match the information listed in the spreadsheet with that particular specimen in the collection. I was not able to enter every specimen into the spreadsheet, but I did learn key characteristics that will help me distinguish solitary bees when I’m out in the field in Minnesota this summer. taxon. I started grouping more recognizable groups together like Agapostemon virescens before looking at more difficult specimen. Once all grouped together, I would enter the information listed on the label into the spreadsheet. I started with the specimen at the top left corner and worked my way to the bottom right corner of the box. I did it this way to make it easier for anybody to match the information listed in the spreadsheet with that particular specimen in the collection. I was not able to enter every specimen into the spreadsheet, but I did learn key characteristics that will help me distinguish solitary bees when I’m out in the field in Minnesota this summer.
Another project that I worked on while at the garden was looking at images of Echinacea styles to see whether or not foreign pollen grains were present. Every style had three images at varying depths. This was done to get a better look at the pollen present (or absent) on the styles. Over the three weeks that I’ve been at the garden I’ve checked 646 styles for foreign pollen and since each style has three different images, I’ve looked at over 1930 images. You may think I’m an expert by now at recognizing foreign pollen, but I’m still very uncertain about what’s present. However, thanks to Tracie, a system was set up to gauge this uncertainty of whether or not there is foreign pollen present. 
Even though it sounds like I spent most of my time in the lab, I was actually outside collecting solitary bees and testing them in emergence traps majority of the day. Once I come into the lab in the morning, I immediately grab the bee-catching net and plastic vials that are in the lab. I also grab my lucky bucket hat before heading out. I head over to the prairie area in the gardens and try to look for areas that have a dense population of golden-rods. When I first started out this summer I had trouble catching bees, but now with a few weeks of experience underneath my belt I’m able to catch solitary bees with and without the net. I’m able to catch the bees without the net by closing the top of the vial around it while it’s resting atop of flowers. Once I’ve caught a few bees (I catch about four to six solitary bees per day) I head back to the lab and grab the emergence traps. I return back to the prairie area and set up a trap on a south facing slope in order to record the behavior of the bee. I either record footage of the bee or record my observations in a notebook, it all depends on how well I’m able to see through the trap. Once I have my observations/film for the day I return back to the lab and share my findings with Stuart.
On a typical day I would be rotating through these three projects, but I’ve also been able to sit in on a few presentations, including a master thesis defense and a PhD seminar. While at the garden I’ve learned many skills that I hope to continue using this summer in Minnesota!
Today will mark the completion of my first work week here at the garden so I just wanted to summarize some of the tasks I’ve been doing for the past five days. The start of the week was dark and gloomy, like any typical Monday, but it was accompanied with a downpour that left me soaked when walking to the Plant Science Center from the train station. Because of the weather I wasn’t able to go out and collect any solitary bees to use in the emergence traps for Kristen’s experiment. Instead I cleaned a few Echinacea heads, identified foreign pollen and continued to develop my skill for identifying the different bees native in Minnesota. Tuesday was a little better. I wasn’t completely soaked, but it was still cold on the walk in that morning. Once again I wasn’t able to go out and collect any bees. However, I was able to identify a large portion of the pollen slides. Also, I found my own method of cleaning Echinacea heads. I use a smaller pair of tweezers to pluck out the achenes and use the larger pair to brush out any that I may have missed. It’s not the fastest way of cleaning, but I think it’s pretty effective.

Wednesday it was finally warm enough to go out and collect solitary bees. My first day out catching solitary bees didn’t go so well because I didn’t catch any. I did catch a few hoover flies and honey bees and recorded their behaviors in the emergence traps. Before leaving that day, Tracie, Kristen and I set up an emergence trap in the prairie area to see if we would catch anything. The next day when I checked the trap we had a few specimen in the trap. They weren’t bees, but now we know that the traps work! Later that day I was able to collect some solitary bees and record their behavior. Today I was able to catch more solitary bees and record their behavior. This time around I decided to film their behavior in the emergence traps using my phone which I will continue to do. I was able to take a video of a Ceratina that I collected, but because the video size is too large I can’t upload it here. However, I was able to upload another recording of an unidentified solitary bee in the emergence trap. I wasn’t able to put the bee into a vial and identify it back in the lab once it crawled from under the trap, but hopefully I’ll catch another one and identify it next week!
Greetings floggers!
The interspecific competition has offered the following results:
- Interspecific competition of growth rate is greater for Bromus kalmii with Elymus canadensis compared to intraspecific competition of Bromus kalmii at this stage of growth.
- The growth rate of interspecific competition for Elymus canadensis with Bromus kalmii demonstrated a negligible change compared to intraspecific competition.
*To view the full write up click here: Interspecific comp.
 Fig 1: Vertical black line on each histogram indicates the mean value for each treatment (E. canadensis vs B. kalmii, E. canadensis vs E. canadensis, B. kalmii vs E. canadensis, or B. kalmii vs B. kalmii) of interspecific competition and intraspecific competition. (F) indicates the focal plant. Likewise, my spring semester internship has concluded as well. This experience has been an amazing and immersive learning opportunity. Within the past week, since the end of my internship, I’ve noticed a shift in the way I think. I’ve started observing plants, not just for their beauty, but also for their seed composition and amazing structures. I have found an appreciation and curiosity for the conservation of our native species and the ecosystems they take part in.
The CBG is a wonderful place that cultivates the excitement of science. Whatever the future holds, I will maintain the same attitude as I have during this experience. From day one, I knew I was in a special place and needed to absorb every moment of it. This internship has truly exceeded my expectations.
In closing, I’d like to thank Stuart and Tracie for their guidance, support, and exposure to a fascinating study of plant science. I also would like to thank them for transplanting my grasses to the prairies of Minnesota, where they can grow to their full potential 🙂
 E. canadensis transplanted to the MN prairie by Tracie Hayes and Stuart Wagenius. Grown from seed by Danielle Oilschlager 🙂 Sincerely,
Danielle
Do you want to germinate some seeds? A while ago Andrea Kramer recommended four resources to find out about seed germination. I’m copying them here:
1. A good place to start with when you are working with a new species: http://www.charlesgwillis.com/baskin-dormancy-database/ This database goes along with the 2014 Baskin seed book, below. It is free but requires sharing your contact info.
2. The book: Baskin, C. and J. Baskin. 2014. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination. 2nd ed. Academic Press, New York.
3. The Royal Botanic Garden Kew has a Seed Information Database: http://data.kew.org/sid/
4. A handy paper with guidelines for a “move-along” experiment: Baskin, C. C., and J. M. Baskin. 2003. When Breaking Seed Dormancy Is a Problem: Try a Move-along Experiment. Native Plants Journal 4:17-21.
Bonus:
5. Prairie Moon Nursery has a Cultural Guide that lists germination requirements for hundreds of Midwestern U.S. species. It’s a big excel spreadsheet that you can access from this page….
https://www.prairiemoon.com/blog/resources-and-information
6. For germinating Echinacea angustifolia, we use a modified version of the protocol developed by Feghahati & Reese in 1994. We don’t use fungicide, but otherwise stick pretty close to their recommendations. If you use this protocol, please cite this paper.
Over the past few weeks, Elymus canadensis and Bromus Kalmii have taken to the plots for a battle of resources. When I initially began this experiment, I hypothesized that Panicum virgatum would take the lead, since it develops a ribosomal root system and is known to be an aggressive prairie species. However, out of 500 seeds, only 1 P. virgatum has germinated and sprouted. For that reason, I’ve decided to eliminate P. virgatum from the data analysis. Although, I have planted the lonely P. virgatum so that it can later be transplanted to the prairies of Minnesota. I later suspected that E. canadensis would take the lead in sprouting. However, B. kalmii has currently overthrown the number of germination/sprouting. There are still a few weeks to go with this experiment before analyzing the data collected on height. With that said, it could go either way. The grasses are growing taller and faster than any of us expected. I take measurements weekly and can already conclude that competition is occurring. The leading height in each treatment varies, indicating that one of the species is achieving more resources than the other. More updates and photos to come!
Sincerely,
Danielle
 Competing species – 2 seedlings to a cell  Bomus Kalmii germinating/sprouting in the agar-petri dish  The one and only germinating/sprouting Panicum virgatum
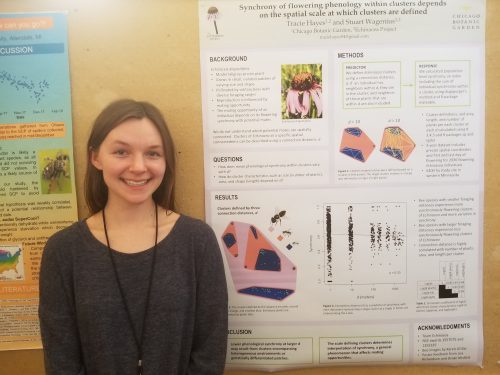
Hi flog! This past weekend I presented my cluster/spatial scale research at the Midwest Ecology and Evolution Conference at the Kellogg Biological Station. Check out my poster!
 Synchrony of flowering phenology within clusters depends on the spatial scale at which clusters are defined – Tracie’s MEEC 2018 poster  Tracie with her poster at MEEC 2018
The seeds were pulled out of their pre-treatment and were moved into the growth chamber for germination to take place. After 5 days, a handful of E.canadensis were germinating, along with B. kalmii at a close behind rate. P. virgatum has yet to germinate, though at this time it’s still early. After filling three 20×10 flats with soilless soil, prior to removing the seeds from cold stratification, the cells (tiny plots on flat) where numbered in preparation of seed transport. By choosing soilless soil I can ensure a uniform system of adequate water retention and proper drainage. As a method for watering, my mentor Stuart has suggested to bottom-water the plants to avoid dampening one area of soil more than another area, which can occur when watering from above. By using sterilized tweezers, I carefully selected a germinating seed from a randomly ordered petri dish. Once the radical was successfully extracted from the agar, the seed was planted into its designated cell. Before transportation took place, I created a few randomizing sheets on an application called R. The sheets randomized the petri dish order, as well as the treatment (species vs. species) placement. The reason we want the order of everything to be randomized is to avoid any biased decisions. This also helps to yield accurate results. I was able to plant over 100 treatments this past week for E. canadensis and B. kalmii. I’m hoping this week P. virgatum will begin germinating.
With that said, the next phase has begun- measurements of growth. To reiterate the purpose of this experiment, I will observe the growth of each species in relation to the species it’s competing with for resources such as water, root space, and light. I will do this by measuring their height on a weekly basis. I want to determine which species will be the most dominant in this early stage of development. On Friday, I started measuring my little sprouts. The tallest I have observed so far has grown to 70 mm. 🙂
This process is very exciting, as I’ve mentioned before. To be able to plan something out so specifically detailed and to watch the process actually happen is nothing short of magical 🙂 It’s really fascinating to watch these little seeds germinate into such beautiful green sprouts!
 Elymus canadensis germinating  Bromus kalmii germinating  Carefully transporting germinated seeds into their randomly assigned cell  sprouting and reaching for light 🙂
Greetings floggers!
This past week steered off the E. angustifolia path for an exciting development on related research. I am beginning my experiment by researching two c3 prairie grasses- Elymus canadensis, Bromus kalmii, and one c4 prairie grass- Panicum virgatum. In the weeks to come, I will randomly plant two differing species in a plot and observe their competition for resources. To begin this process, I have prepared a pre-treatment of cold-stratification to help break seed dormancy. Pre-treatment consisted of filling fifteen 150 x 50mm petri dishes with agar, bleach treating seeds (to prevent growth of any possible bacteria or fungi), plating the seeds on the solidified agar, and refrigerating for two weeks. Each species filled five petri-dishes (1oo seeds per dish). After their pre-treatment of cold stratification, the seeds will stay in their dishes and be transferred to our grow chamber for germination. Once germination is observed, they will find their home in the soil. 🙂 This is very much a summery of the experimental process. However, it’s exciting news and relates to the Echinacea project on the grounds of prairie restoration. Upon completion of the experiment, the prairie grasses will be transported to our common gardens in Minnesota, which each species happens to be native to. Stay tuned for more exciting updates!
Sincerely,
Danielle
 Weighing out agar
 Heating agar mixed with deionized water  Agar solidifying into gel under UV light to assist in sterilization  Moving seeds (after bleach treatment) to petri dish on top of agar gel  From left to right- B. kalmii, E. canadensis, and P. virgatum – ready for cold-stratification in the refrigerator
Here are the books I recommended in today’s professional development seminar on Science Communication for the PBC graduate program. Best wishes in your endeavors to communicate more effectively! Everyone benefits when you communicate better, especially you.
Carol S. Dweck. 2006. Mindset : the new psychology of success. Random House.
Daniel Kahneman. 2011. Thinking, fast and slow. Farrar, Straus and Giroux.
Stephen B. Heard. 2016. The Scientist’s Guide to Writing. Princeton U Press.
Heath & Heath. 2007. Made to Stick. Random House.
Randy Olson. 2015. Houston, we have a narrative: why science needs a story. University of Chicago Press.
Anders Ericsson & Robert Pool. 2016. Peak: Secrets from the New Science of Expertise. Houghton Mifflin.
I also mentioned SMART goals. Wikipedia reveals all you need to know.
In a previous flog post, I recommended several books about managing your time.
|
|


 taxon. I started grouping more recognizable groups together like Agapostemon virescens before looking at more difficult specimen. Once all grouped together, I would enter the information listed on the label into the spreadsheet. I started with the specimen at the top left corner and worked my way to the bottom right corner of the box. I did it this way to make it easier for anybody to match the information listed in the spreadsheet with that particular specimen in the collection. I was not able to enter every specimen into the spreadsheet, but I did learn key characteristics that will help me distinguish solitary bees when I’m out in the field in Minnesota this summer.
taxon. I started grouping more recognizable groups together like Agapostemon virescens before looking at more difficult specimen. Once all grouped together, I would enter the information listed on the label into the spreadsheet. I started with the specimen at the top left corner and worked my way to the bottom right corner of the box. I did it this way to make it easier for anybody to match the information listed in the spreadsheet with that particular specimen in the collection. I was not able to enter every specimen into the spreadsheet, but I did learn key characteristics that will help me distinguish solitary bees when I’m out in the field in Minnesota this summer.