|
|
Hello! My name is Emma Carlson, and I am one of the Lake Forest College student interns working at the CBG! I am currently working with Tracie Hayes and Stuart Wagenius on the aphid exclusion and addition project. Over the past few weeks, I have been working on cleaning Echinacea heads, as well as randomizing and counting achenes! I have thoroughly enjoyed being a part of this project and being able to actually DO science! As a future science teacher, I always want to emphasize to my students that science is not just something you learn or read about, but something that you do and engage in! Science is about exploration, discovery, research, and hands-on problem solving. I am LOVING this opportunity to get out of the classroom and actually do science with this internship!
I am so thankful for this opportunity to see and experience how all of the different parts of the project, such as counting thousands of achenes, contributes to the overall importance of the study. This mini-internship has been an extremely valuable experience for me, and something that I am honored to be a part of!
Here is a few photos of me, working super diligently to beat Leah’s overall achene count for the day!


 Anne has counted over half a million achenes! Today we celebrated Anne’s accomplishment of counting over 500,000 achenes with a prairie remnant made out of cupcakes. Anne has been a member of Team Echinacea for over 10 years and she has really put in the hours! We can’t thank her enough; it’s great having her in every Friday.
Notice the cupcake “soil” under the diverse cupcake prairie remnant – complete with Echinacea, Helianthus, bottle gentian, and grass!
 Prairie cupcake remnant. Are these bare ground cupcakes good for solitary bees??
 Prairie cupcakes detail.
Stuart, Michael and I were back in Minnesota this week Monday-Thursday to finish up a few fieldwork tasks. We staked to 268 points across our sites to complete demo rechecks. Most of our Echinacea have fully senesced, but there were a few out there with some greenish leaves. There are not a lot of species flowering, except your occasional Solidago still holding on.
We also started work herbiciding Ash trees in p8. We didn’t apply herbicide to any trees close to Echinacea, because we wanted to test out our methods. It will be interesting to see how well the herbicide works. Finally, no chances for a burn these last few days, with wind and humidity levels not right. There might be a chance for a burn this fall, but more likely spring 2019.
 Solidago fluff at Hegg.
Your photo-blog-flog fix.
 sling attack at eri this morning  Artemisia frigida at Loeffler’s Corner West!  late bloomin’ liatris at lce  Serendipity! Nessman sling!  Before and after: I harvested a 13 + 1 headed plant at p2 today!  The beautiful late-season walk to p2! Sorry the daily flogging has been irregular these last few days. Since our last entry, we’ve been harvesting Echinacea from p1 and p2, sling-ing, p8 rechecks, and field checks. Lea has been working on demo, phenology, and surveying nearest neighbors along her spp transect, and Kristen has been moving traps and measuring soil and vegetation. We’ve been busy!
I’m so happy to be back in MN as our fieldwork for 2018 finishes up! There has been plenty to do, especially with harvest gearing up. We started out the morning finishing flagging p8 so that it will be easy to avoid Echinacea when we want to spray the Ash trees. Then, after a couple rows of harvesting p1, we had an early lunch so we could go help Kristen take down her emergence tents. We finished the day off with a long session of harvesting p2. A majority of the heads are ready for harvest, and a lot of them have lost achenes/entire heads, likely due to predation or the storm on Monday.
 View of P8 from row 19, with Andy harvesting in the distance. I haven’t been here since burning in early spring and I’ve really missed it. Muscle memory has kicked in and I am getting into the rhythm of fieldwork again. Excited to be back and experiencing the late season prairie again!
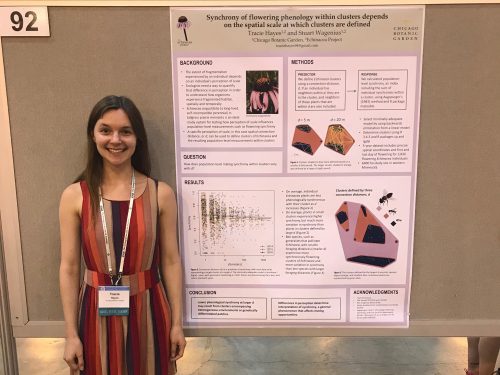
Last week, I attended ESA for the first time and presented a poster on a project I’ve been working on for the past few months: how the synchrony of flowering phenology within clusters of Echinacea depends on the connection distance used to define those clusters. I presented on Tuesday, August 7, 2018 in PS 18: Habitat Structure, Fragmentation, Connectivity from 4:30-6:30, board #92 (just feet away from Will’s poster). My main results are that clusters of Echinacea defined by a small connection distance tend to have lower synchrony on average than clusters defined by larger connection distances. Clusters defined by smaller connection distances also have more variation in synchrony. In terms of a bee’s perspective, this could mean that bees with smaller foraging distances are experiencing more synchronous clusters of Echinacea as they travel from one plant to the next. However, the experience from one small bee to the next is variable. Larger bees with larger foraging distances might be experiencing clusters that are more asynchronous, so as they travel from one Echinacea to the next, plant flowering times might not be overlapping as much.
There was an almost continuous flux of people coming by, and even though I was nervous at first, these couple of hours were probably my favorite part of the conference. Even if some of the listeners didn’t ask me specific questions at the end, just describing my project over and over made me realize what parts I wanted to continue thinking about and working on. I had scientists come by that I recognized from talks I had seen, Team Echinacea alumni interested in what we are doing now, and people I didn’t know that just came because of the title! It was all really exciting and I have a page of notes with questions and ideas to think about as I move forward with this project.
The conference as a whole was a really great experience for me, because I could start to see how both this specific project and my general interests fit in with the rest of the ecology world. It helped me to start to define the questions I want to ask as I think about grad school and the future.
 Tracie and her poster at ESA 2018 🙂 Stay posted for more updates on this clusters project!
Link to PDF of Tracie’s ESA poster
Last week, Allen and his wife Doris won an amazing award at the Volunteer Awards Reception! Congratulations Allen and Doris!
Allen and Doris were presented with the 2018 Kris S. Jarantoski Excellence In Horticulture Volunteer Service Award, an award that recognizes volunteers for “their dedication to nature, enthusiasm for education, and exemplary volunteer service in support of horticulture and plant collections at the Chicago Botanic Garden.”
We have been so happy to have Allen on our team. He is a long-term volunteer at the garden and has worked on a variety of different projects over the years. He comes in on Tuesday afternoons to work with Team Echinacea and is one of our critical achene counters! Thanks for all of your hard work, Allen- this award is well-deserved!
 Allen counting achenes.
Susie, a member of Team Echinacea since 2011, was also recognized for volunteering over 600 hours this past year! That is quite an achievement. Susie would come in all day Tuesdays and Thursdays, starting out with randomizing or cleaning in the mornings and then working in the herbarium in the afternoon. Susie did so much great work for the Echinacea Project, always had a smile, and was always keen to learn about what the lab managers and grad students were working on.
 Susie with all of her randomizing materials out.
During the summer of 2017, Lea and I carried out a project we called “Rich Hood” (richness of floral neighborhoods). This involved setting up 2 x 2 m square plots around Echinacea plants in the remnants, and getting cover class estimations for all species present. We also harvested flowering Echinacea heads, and there is more specific information in the flog post about reproductive fitness in the remnants. Nina Denne, a student at Carleton, completed an externship project comparing the floral neighborhood with the seed set of the collected Echinacea heads.
On May 6th and May 7th, 2018, I returned achenes to the remnants that were not sampled for the x-ray. I returned achenes to all the remnants that were in our study except for Landfill and SPP, which will hopefully be rematriated later this spring. At each site, I staked to points where heads had been collected (using stake file stakeReturnRichHood.csv), found the matching tags within the plot, and spread achenes in about a 20 cm radius around last year’s stalk. In some cases, if I couldn’t find the tag within a reasonable time of searching, I spread the achenes around the point I staked to.
It was nice to see what the remnants looked like in the spring, but I didn’t see any tiny Echinacea rosettes yet. Some of last year’s heads out there had dispersed all of their achenes, but many were still holding on to a few.
 An Echinacea head with a few achenes left to disperse.
Hi all! Stuart and I are in the process of figuring out the best way to edit our x-ray images of Echinacea achenes so that it is easy to classify them:
- Are all achenes (full and empty) visible?
- Is it easy to tell between full and empty achenes?
- Can achenes within clumps be identified individually?
 Example x-ray image with achenes, before edited Here is an example of an x-ray image that is tricky to classify. All of these achenes are empty, so they are more translucent and harder to see. Also, many of these achenes are in clumps, so getting the correct count of empty achenes is a challenge.
 Example x-ray image before edits, with circled clump A single clump has been circled in green in this image. This clump has 3 empty achenes, but we want that to be more obvious. To edit our images, we have been working with the EBImage package in R. Here are a few examples.
 Edited example #1 This edit is helpful because it makes the background darker so that it’s easier to pick out achenes. The 3 in the clump are still kind of hard to pick out, though. Here is the code we used:
library(EBImage)
x <- readImage('https://echinaceaproject.org/wp-content/uploads/2018/04/x2mid.jpg')
display(x)
kern = 2000*makeBrush(99, shape = 'Gaussian', sigma = 5)
display(whiteTopHat(x, kern))

This example is blurred a bit, which is helpful for seeing achenes in their entirety. This is the code we used:
library(EBImage)
x <- readImage('https://echinaceaproject.org/wp-content/uploads/2018/04/x2mid.jpg')
display(x)
display(gblur(x, sigma=0.8))
There are endless possibilities if using multiple EBImage functions on one image, and we are looking for some guidance. If you have experience with EBImage, what do you think are the best combinations of functions for reaching our goals? Thanks for the help!
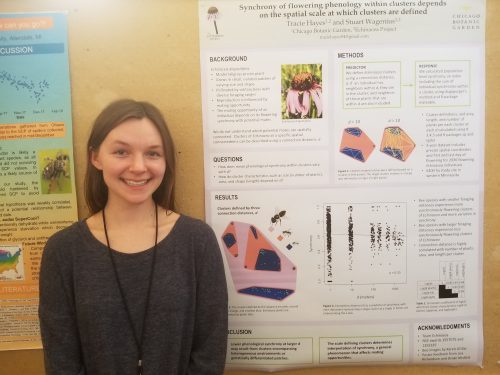
Hi flog! This past weekend I presented my cluster/spatial scale research at the Midwest Ecology and Evolution Conference at the Kellogg Biological Station. Check out my poster!
 Synchrony of flowering phenology within clusters depends on the spatial scale at which clusters are defined – Tracie’s MEEC 2018 poster  Tracie with her poster at MEEC 2018
|
|