|
|
Hi everyone! Tracie, Kris (another PBC grad student), and I had a great time presenting this year at the Midwest Ecology and Evolution Conference in Kalamazoo, MI. Here’s a look at my poster that I presented about pollen on Echinacea as a part of the ongoing Floral Neighborhood Communities project:
 Do pollen loads differ among native bee visitors to Echinacea angustifolia?  A picture of me presenting! MEEC 2018 was awesome! For any undergrad or graduate student interested in attending an inexpensive, regional conference I would highly recommend it. It was great to network with fellow graduate students and hear all about the great research ongoing here in the region!
Hello friends,
We just finished a rousing lunchtime discussion on the virtues of archiving, so I thought this might be an appropriate time to post our compiled data set from all four days of pollinator observations and captures.
compiledpollenobs2009.xls
Here are files with presence/absence of species within the 2m floral neighborhoods
fnc10mNeighborhood.csv and within 10m floral neighborhoods fnc2mNeighborhood.csv. The first is just a reorganization of FNC.csv and the second includes info from FNC.csv and from WITHIN10M.csv.
Listen up, Echinacea fans!
I’ve now finished making slides and taking photos of the first 68 insect visitors–only 107 left to go.
Here are some photos of the process:
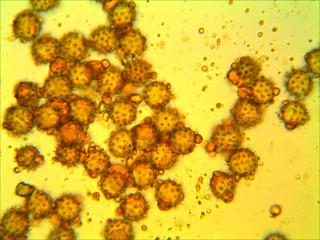
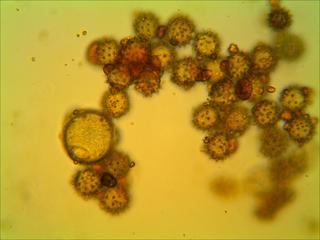
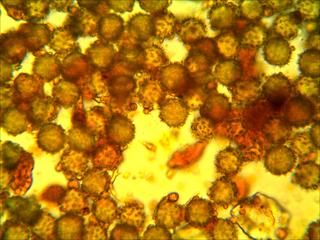
1) Here is an insect carrying LOADS of pollen (haha! get it?) which I am about to transfer onto a small agar cube on a microscope slide.

2) I heat the slide, complete with pollen-covered agar cube and cover slip, on a hotplate. Next I throw the completed, labeled slide under the microscope camera and take photos like these : :


3) I’ve pinned each specimen with a unique ID code that corresponds to its vial ID number.


The most common genera near as I can tell from the reference collection are…
Male Melissodes sp.

And Ceratina sp.

Please leave questions or comments and I’ll do my best to respond!
-Amanda
So, I’ve been working hard creating slides from all the styles everyone helped to collect (thanks!), and this is the protocol I’ve been using:
II. SLIDE PROTOCOL:
A. ORGANIZE THE STYLE VIALS BY SITE
B. CREATE SLIDE LABELS
C. RANDOMIZE THE STYLE INFORMATION ON EXCEL
D. CREATE THE SLIDES IN THE RANDOM ORDER GIVEN BY EXCEL
1. Place the blank slide on a clean surface
2. Pull out the correct vial, open carefully (sometimes the styles are on the lid of the vial), use clean tweezers to remove the contents. Place vial contents onto the slide.
3. Remove any anther parts from the slide; organize the stigmas so they are separate/easily differentiated from each other.
4. Place one drop of glycerin on top of the stigmas. If there are any bubbles, try to move them away from the stigmas.
5. Place a cover slip carefully over the glycerin and allow it to settle.
6. Use mounting medium to seal the cover slip to the slide. Allow the slide to dry on a flat surface for 24-48 hours.
E. Put completed slides into slide boxes.
Of the approximately 380 slides I started with, I have 150 left to do, so I’m more than 1/2 way there. Once I’ve created all these slides, I’ll start taking photos and uploading pictures.
Anyway, just felt like it was about time I updated. Gonna go make slides now… ^_^
Here is the data that’s been compiled for FNC, pollinator observations, within 10 m, and the isolation measure of flowering Echinacea focal plants.
Some things I wanted to point out that may or may not make a difference:
>In the FNC data, all quadrants with none present have no data for distances, and sometimes there is only one distance if there was only 1 infl of a species within a quadrant…
>I still need to check each tag number that we recorded for FNC and make sure it coincides with the original record of tags we made when we flagged.
>For the isolation measure, I put in 11 for distances >10m. I marked the original distance in the notes in cases where they were >10m but measured out (i.e.SPP)
>For inflorescence counts>100, I put in 1000. In the comparison file below, we changed 1000 to 101 since we had to sum the inflct for each unique ID and so some of the infl ct were showing up as >1000.
ISOLATION.csv
FNC.csv
POLLOBSWITHFLIES.csv
POLLOBSWITHOUTFLIES.csv
WITHIN10M.csv
pollcomparison ecan mesa amca.csv
ech mesa comparison with mean std error.csv
ech amca comparison with mean sterror.csv
As of 8/3:The last 2 files are new. In order to make the graph that appears on my poster, we divided the unique Id’s into 4 groups: 1-alfalfa only in fl.neighborhood 2-ech only 3-both 4-neither. I took the average and standard error from each of those 4 groups to make the 4 bars on the graph. I want to do the same thing for Amorpha. So I attached the file that I’d use to make the same graph but with amorpha. You could use the third to last file which has infl ct for each unique ID for ecan, mesa, and amca to do the analysis but I figured I’d put the others up so you could know how I made the graph…
FYI, after Amanda and I looked at all the vials yesterday, it turns out that over our 4 day stint we had 132 bees, 13 scsf, 33 flies, 4 butterflies, 15 beetles. Good work everyone!
This two-part entry includes one observation about pollination that struck me odd. I had a floral head (A) at Loeffler’s corner and as Agopostamas texanus approached – it stopped – flew backwards and away – and visited others nearby (all Echinacea). Did the presence of ants on the head – around the anthers have anything to do with the “I’ll just come back later” actions of the bee? Has anyone else observed a head NOT get visited even though it was ripe with pollen because of the presence of ants?
My second half is simply noting that a calico cat and two large kittens were at the end of the common garden yesterday as I left about 4PM. The mother slunk away and the gold/white kitten watched me while the other kitten mostly white/ some black was trying to consume a chipmunk! Are these cats known to inhabit the area?
Gretel- KJ’s
Mimi- Railroad Crossing
Allegra- Landfill East
Stuart- Staffanson
Amy- Steven’s Approach
Amanda- Yellow Orchid Hill
Daniel- Aanenson
Kate- Riley
Caroline- On 27
Greg- Loeffler’s Corner
See you guys at 7:30ish at Hjelm House!
xoxo,
Amanda
We have made a few changes to the protocol for tomorrow’s FINAL outing for pollinator observation and collection:
1) DO NOT capture syrphid flies!
2) If you observe a small common syrphid fly (Family: Syrphidae, Sphaerophoria sp.) on your Echinacea head, make a note that one SCSF was observed. Here are some photos of syrphid flies:
http://echinacea.umn.edu/insects/images/poll2005vin1948side.jpg
http://echinacea.umn.edu/insects/images/poll2005vin1927top.jpg
http://echinacea.umn.edu/insects/images/poll2005vin1927front.jpg
Here’s a video that might help:
http://www.youtube.com/watch?v=UkjPfAMGmiw&NR=1
———————————————————————————————
As for pollinator collection and analysis…
We’ve gone out collecting three times now (tomorrow is the fourth and final day), and we have about 140 Echinacea insect visitors in vials in the freezer. Here is my protocol for making slides and labeling specimens:
1) Remove vial from the freezer and allow insect to thaw
2) Cut a very small (2mm cubed) piece of agar and situate it in the middle of a slide.
3) Remove the insect from the tube and dab and sweep every surface of the body across the agar cube. This is intended to simulate the amount of pollen that might be transferred to Echinacea styles during a visit.
4) Set the insect aside on Styrofoam.
5) Sweep the inside of the vial with the agar cube for any loose pollen.
6) Place a cover slip atop the agar cube and put the slide on the edge of a hotplate on LOW heat. Watch the slide carefully and remove it when the agar is soft but not melted (once melting starts, it will quickly boil and create bubbles in the slide).
7) Pin the insect (for identification if it is an unknown species, or quick-n-casual for a known species) and pin it with a new one letter, three number code.
8) Label the slide with the vial code and the specimen code.
Photos:
I photograph the slides on the same day as I make them so that the agar doesn’t dry out before the photo. To sample the slides, I have generated random pairs of numbers from 1 to 22 (inclusive). The cover slips are 22 x 22 mm and I use the ruler on the microscope to scroll to the randomly selected “plots” from one corner of the cover slip. From there, I take the photo at 40x. I take ten photos for each slide, and sometimes take more than one photo per plot if I capturing all of the pollen grains requires multiple focuses.
I will post some photos of this process soon, so hang in there!
As always, feel free to leave questions or comments.
Thanks!!
Amanda
Kate and I made a list of species coflowering with Echinacea at Ri, LC, and rrx yesterday. These species are not within the 2m floral neighborhood, but are within 10 m of at least one of the observed plants. coflsp13jul2009.xls
|
|