|
|
I have some pictures of pollinators, but more to choose from would always be helpful for putting together my poster in a couple weeks.
I know Gretel and Josh said they had some, anyone else?
If so.. I would like any good pictures of Melissodes, Agopostemon, Augochlorella, Ceratina, and Lasioglossum. Preferably on Echinacea.
If you could go through them and put them on a USB for me, that would be greatly appreciated. And I would acknowledge you for the picture if I use one of yours.
Thanks,
Katie
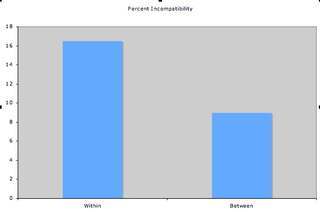
After a couple of weeks of work 278 crosses in the common garden have been completed. Attached is a csv file containing the data collected. I have yet to finish collecting dates of the end of phenology for some of the plants so once all flowering is done I will post the remaining data. I ran a chi-square on the between and within crosses for compatibility and came up with a nearly significant result (X2 = 3.62, df = 1, P = 0.057). Below is a graph representing the difference in compatibility for between and within remnant crosses. As expected, there was higher incompatibility for within remnant crosses than for between remnant crosses. I will continue to post statistical results along with project photos and methods. Please feel free to question or comment.

btw.within.crosses.csv
In case we need help 🙂
Materials:
Visor
Petri dish
paintbrush
mesh bags
twist ties
a partner!
1. Sync visor, get randomized plant (row and position) from Hillary.
2. Collect aphids from several plants in Common Garden. Find leaves with fat, dark aphids (aka mature). Gently disturb aphids with brush tip. When aphids start to scurry (remove stylets), brush them gently into the petri dish. Do not mash them by rubbing paintbrush against the plastic (attempting to dislodge them).
3. When approximately 20 mature aphids have been collected (not including tiny green ones), find assigned plant.
4. Check transfer plant for ants and aphids and record presence in form.
5. Find a suitable leaf (close to ground and small enough to fit in bag) that is free of any ants and aphids (if there are no empty leaves, squish present aphids).
6. Prepare bag over most of the leaf (opening bag and pulling over leaf). Then lift the bag up enough to stick the aphids in.
7. Transfer two random aphids to the top of the leaf (one big aphid and one slightly smaller). It works best if one partner holds the leaf and bag and the other transfers the aphids. Then the bag holder pulls the bag down and the transferrer twists the tie.
8. Pull bag completely over leaf and gently twist tie it closed. Avoid strangling the leaf or squishing or dislodging the aphids.
9. If aphids take a death plunge or fall off make sure they are not in the bag and transfer additional aphids as needed.
So far the phenology is going good, Heliopsis seems to be finishing up flowering, and I was able to pull some flags today! All the echinacea and coreopsis seem to be just at peak or a little past, while the carduss and flags are continuing to be mowed. I am starting to input my data and will probably have some preliminary graphs up next week!
The string segments that came with the wheeled trimmer were 47 cm long. I used 32 cm sections for trimming rows in the CG.
It’s been quite a while so there is lots of good news on the aphid front! First of all, aphid casualties have decreased substantially thanks to improved transfer methods. We have now been using a paint brush to gently disturb and brush the aphids into a petri dish (upon the suggestion of Dr. George E Heimpel). This is far less traumatizing for the aphids and survival has skyrocketed from 20% to around 50%!
Also, fortunately for us, apparently all aphids at this time of the year are gravid, so we only need to select mature individuals for transferring. This was an exciting discovery since we spent a while with a microscope trying to figure out which ones were gravid before this revelation. We also officially decided to use two founders for each transfer to improve chances of survival.
Hillary and I are looking forward to checking out the preliminary data soon!
 Beetles getting busy. The male was mounted on the other female shortly before this photo. Beetles getting busy. The male was mounted on the other female shortly before this photo.
 Looking for orchids, this is in some pretty swampy area. Someone Mentha. Looking for orchids, this is in some pretty swampy area. Someone Mentha.
 Swamp milkweed, Asclepias incarnata. Also while out looking at orchids. Swamp milkweed, Asclepias incarnata. Also while out looking at orchids.
 Showy milkweed, Asclepias speciosa. Showy milkweed, Asclepias speciosa.
 Lepidopteran love. Lepidopteran love.
 This plant appears to not be photosynthetic This plant appears to not be photosynthetic
 Click to embiggen. This white fuzzy (yes, an insect!) is hanging out on an echinacea, doing whatever it is they do. Click to embiggen. This white fuzzy (yes, an insect!) is hanging out on an echinacea, doing whatever it is they do.
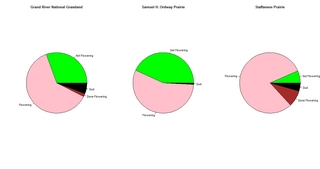
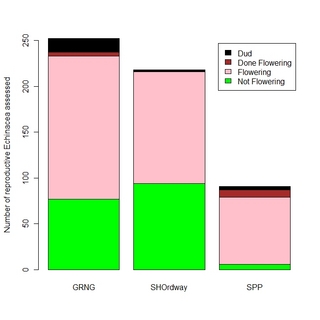
Last week I assessed Echinacea flowering phenology at Grand River National Grassland south of Lemmon, SD, Samuel H. Ordway Prairie west of Leola, SD and Staffanson Prairie near Kensington, MN. Here are a couple of figures I generated to compare phenology at the 3 sites.
First, I made pie charts to show the relative proportions of flowering plants.

Next, to show more quantitative information, I used a stacked bar graph.

These figures illustrate that the flowering phenology is most advanced at Staffanson and least advanced at S. H. Ordway Prairie. Nevertheless, I am encouraged that there are lots of flowering plants at all 3 sites, suggesting that a long-distance cross involving plants from these 3 locations would be possible. I am considering tackling that project next summer, to assess whether there would be lower seedling recruitment from between-population crosses compared to within-population crosses at these 3 sites.
Here’s a picture of some flowering Echinacea at Perch Lake, which is near the S. H. Ordway prairie.

Here is a picture of 2 of the 3 surviving plants in my experimental plot at Perch Lake Waterfowl Production Area, southwest of Leola, SD.
 Each of the 2 Echinacea seedlings is marked with a red party toothpick. We (Shelby, Janelle and I) found them in May when we censused my 2 SoDak plots. We found a total of 10 new seedlings at the Perch Lake site, all of which had 2 cotyledons but no true leaves. I returned to Perch Lake last Thursday, July 8. Sadly, 7 of the seedlings were gone, but I was able to verify that the 3 survivors are, indeed, Echinacea angustifolia. Each of the 2 Echinacea seedlings is marked with a red party toothpick. We (Shelby, Janelle and I) found them in May when we censused my 2 SoDak plots. We found a total of 10 new seedlings at the Perch Lake site, all of which had 2 cotyledons but no true leaves. I returned to Perch Lake last Thursday, July 8. Sadly, 7 of the seedlings were gone, but I was able to verify that the 3 survivors are, indeed, Echinacea angustifolia.
The Perch Lake site is 1 of 3 experimental plots I (and my assistants) sowed in November 2008, to ask whether Echinacea from western South Dakota, central South Dakota and Minnesota exhibit local adaptation in seedling recruitment. More background and results from the 1st 2009 census are displayed in a poster that you can find in the September 2009 archives of the FLOG.
Unfortunately, the Perch Lake site was sprayed with a combination of herbicides (Tordon and Telar) in August 2009. The treatment was lethal to ALL the Echinacea seedlings that emerged in 2009. Thus, the 3 surviving seedlings from this year are the only living Echinacea in this plot! Fortunately, I was able to census the plot shortly after it was sprayed, and I am confident that I found the survivors up to that point.
I plan to present a talk about my local adaptation experiment at the North American Prairie conference in August.
Today we will start measuring Echinacea in the Common Garden. Here is the link to the protocol: CGmeasureprotocol2010.htm
|
|