Below is one of the scans of Stipa seeds collected from Douglas County. This particular set of seeds was collected from a plant at Staffenson Prairie Preserve. The seeds themselves are pointed towards the left side of the image, and extending from them are the long awns that give Stipa it’s common name (porcupine grass). At this stage, they look less like quills, though, because they have dried and started to coil (click on the picture to see it full resolution and you can actually observe the coils and lots of other neat features of the seed, like hair and a dagger tip!). Out in nature, the coiling action would allow the seeds to attach to a disperser or to drill themselves into the ground in preparation for overwintering and germinating the next spring. We’ll be scanning all of the seeds we collected (an estimated 3,000+ seeds from 431 plants), making digital measurements of seed length and width, and planting around 2,500 of them interspersed with Echinacea in the common garden.
|
||||
|
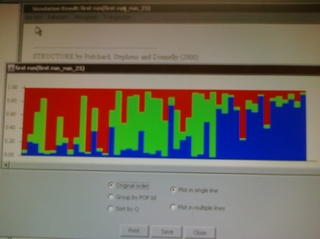
Hi, Team Echinacea — Its Diedre and Jake with an update from the lab at the Chicago Botanic Garden. In spite of several setbacks, including a crowded lab and a power failure that shut the lab down for an entire day, we’ve been able to create a lot of data. Currently, we have ten microsatellite primers working which we use for paternity and genetic diversity analysis. Recently we’ve been able to up our extractions to over 150 samples a week and 10 PCR’s a day! Jake and I started extracting the samples that Jennifer and I took in Minnesota several weeks ago. Jake is using these for his poster on population structure. He will be looking at whether there is interbreeding or inbreeding among the nine remenant populations we sampled (East of Riley, Anenson, Steven’s Approach, Landfill, Railroad Crossing, Staphenson Prairie Preserve, KJ, and Ness). Jake has already found some interesting results with the use of Structure and FStat. The poster I am working on will look at the diversity of pollen donors with regard to flowering on individual and population levels. Here are some pictures of our work in the lab: Jake and I making DNA extractions a little more fun than they already are through use of our artistic talents. Some preliminary results for Jake’s project. We are going to plant Stipa spartea seeds into the common garden. Here is a Here’s some of the work I’ve done with organizing my data. I still need to figure out how to organize it to be able to analyze it, so this is mostly just preliminary work. I have about 2 weeks to put this all together….any help/advice is appreciated because right now, the data I have is a little overwhelming. There are 3 sheets in this document. For next week, it looks like the weather should hold up for Tues and Thurs to be able to do pollinator observations. So we will need to flag the sites on Monday and have everything ready to go for Tuesday. Remember, you ALWAYS record something for each observation you make, regardless of whether or not you observed/caught a pollinator. Select No for poll. observed and No for pollinator caught if this is the case. Some things I wanted to clear up for people helping with FNC: Here’s some of the pollinators I saw on Coreopsis near Hegg Lake. They seemed to only be pollinating Coreopsis although there were other species like Achillea, Amorpha, and Echinacea around. On Friday all of us except Greg went on a trip to a mesic prairie 3 hours away to help Gretel look for orchids. We split into 3 groups of 3 and flagged the flowering plants within the various treatment grids. To celebrate peak flowering in the common garden, Megan made these awesome cupcakes. We all enjoyed them. Thanks, Megan!! Megan with her breathtaking display of deliciousness.
Do you notice the stages of flowering presented here? Mimi contemplates consuming something so beautiful.
Stuart basks in the celebration of his beloved study species.
Daniel enjoys with passion.
Please review this protocol for measuring plants in the common garden. Kate and I made a list of species coflowering with Echinacea at Ri, LC, and rrx yesterday. These species are not within the 2m floral neighborhood, but are within 10 m of at least one of the observed plants. coflsp13jul2009.xls |
||||
|
© 2026 The Echinacea Project - All Rights Reserved - Log in Powered by WordPress & Atahualpa |
||||