|
|
This summer, REU student Jill Gall collected ants from prairie remnants in Douglas County. Part of her project was identifying specimens to genus and sorting them by morphospecies (i.e. you know they’re different species, but you don’t know which species they are). This fall I met with Dr. Sean Menke, an ant ecologist at Lakeforest College, to look at the specimens she collected and get advice about how to identify ants. His advice has helped me make move forward in identifying the specimens we collected this summer.
The challenge with distinguishing ant species is that a lot of the most obvious characteristics, such as size and color, vary within species. The traits that do distinguish species vary across genera. For instance, in the genus Formica, species can be discerned based on the hairs covering certain parts of the body and the glossiness of certain body parts. In Lasius, one of the key traits is the orientation of the hairs on the antennal scape (the part of the antenna closest to the head). With some genera, species identification is especially difficult. One of these is the genus Myrmica.
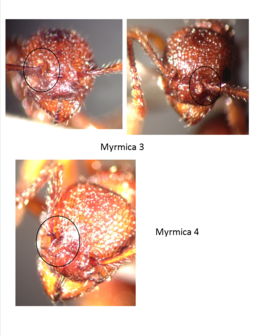
After looking through a large number of specimens, I have come across four types that I believe are separate species. The differences are in the shape of the antennal bend and in the shape of the flange that covers the base of the antennae. I have circled this area in the pictures below. For three of the four morphospecies, I have included pictures of two individuals. See if you can spot the differences.


This week has involved a combination of new projects and ongoing endeavors.
First of all, I should mention that the Echinacea Project is hiring field interns for the summer of 2013. See https://echinaceaproject.org/opportunities/ for more information about job openings.
The volunteers have continued making progress in processing Echinacea heads harvested in 2011. We are nearly halfway through weighing achenes from an experiment planted in 1999 and have have obtained achene counts for 3 of the 9 experiments in the main experimental plot. The volunteers are working on counting achenes for the remaining experiments and on cleaning Echinacea heads harvested in 2012.
More projects has meant more people in the lab. On Thursday, we had a vibrant crowd of five volunteers and two student interns.

One of these interns (not shown in the picture) is Jill Pastick, a junior at Lakeforest college who will be working in the lab until May. Her project will involve germinating achenes from one or two ongoing experiments. One of these experiments is examining hybridization between two species of Echinacea: the native E. angustifolia (the one we work on) and the non-native E. pallida. This summer, Shona Sanford-Long (a junior at Middlebury College) performed experimental crosses of E. angustifolia and E. pallida growing in a Minnesota prairie restoration and is currently analyzing her results to assess the success of different parental combinations in producing fertile achenes. The other experiment examines the effects of burning on offspring fitness in E. angustifolia. Last summer, Kelly Kapsar (a junior at Carleton College) recorded the flowering phenology of Echinacea in burned and unburned units of a prairie preserve. Several interns have been involved in cleaning and weighing the achenes from those plants.The next step in both of these experiments will be to germinate the achenes in the lab and plant them in an experimental plot in Minnesota.
15 January 2013 Amy Dykstra defended her doctoral dissertation, entitled: Seedling recruitment in fragmented populations of Echinacea angustifolia and passed with flying colors! Members of the committee referred to her research as extremely impressive and highly valuable to restoration biologists, among others. At the end of January, Amy will return to Bethel University, where she has been teaching since August 2011.
We are launching into the new year like an Echinacea achene launching from its receptacle. The reason I mention this terrible simile is that we are in the process of extracting achenes from Echinacea heads harvested in 2012. Even though we haven’t cleaned many heads, we have found quite a few stray achenes around the lab–on the floor, in drawer handles, in people’s clothes. One of our volunteers, Lou, developed a device to minimize achene loss. Lou is a mathematician and avid hobbyist engineer who has contributed his talents to various aspects of the project, including building a tray for scanning achenes and a frame for mapping and finding seedlings in the field. Volunteers Bill and Leslie tried out his prototype cleaning shield on Thursday and Friday:


In other news, we are in the process of developing a new system for collecting achene count data. Basically, instead of writing down the count on a paper data sheet, each counter would enter the information into an online form, which would feed directly into the database. Bianca Rosenbaum, the CBG web developer, is helping us develop this system. If all goes well, we should be able to try it out in the next few months.
There is a lot of work left to do for the 2011 Echinacea harvest, including counting achenes on scans and selecting a random sample to weigh. Thankfully, many of our volunteers have returned from holiday vacations and have resumed their invaluable work in the lab. For those that are still in warmer climes, we look forward to their return.
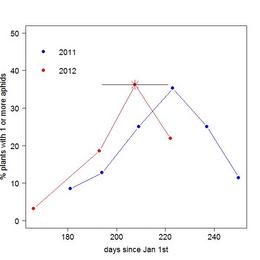
During the summers of 2011 and 2012, I conducted a survey of aphid infestation in a section of the main experimental plot to track, among other things, seasonal changes in the distribution and abundance of Aphis echinaceae on Echinacea. With help from members of team Echinacea, I conducted 6 bi-weekly surveys in 2011 and 3 monthly surveys in 2012. In both years there was a sharp rise and fall in the frequency of aphid infestation. The plot below shows the percentage of plants in the survey area observed to have 1 or more aphids on each dates. In 2011, the peak of aphid infestation (i.e. when the highest percentage of plants hosted aphids) was around August 12th. In 2012, the peak date of aphid infestation occurred some time between July 13th and August 9th. I was not able to observe the peak directly due to a sudden die-off of aphids before my third survey. I estimated the peak frequency of aphids as the percentage of plants with live or dead aphids on August 10th (indicated by the asterix and x-error bar).

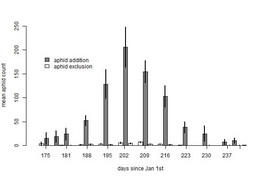
I was also able to get a sense of aphid phenology from my aphid addition/exclusion experiment. In 2011 and 2012, I added or excluded aphids from 100 plants that were not flowering in 2011. The graph below shows the mean abundance of aphids in each experimental group over the summer of 2012:

Based on this graph, I estimate that the peak of aphid infestation for 2012 occurred around July 22nd (202 days after January 1st), about two weeks before last year’s peak.
Today volunteers Bill and Bob spent the morning respectively cleaning and counting Echinacea achenes. Bill (pictured below) came across a receptacle that had a big hole in it–probably due to a burrowing critter:


In other news, Shona Sanford-Long, one of the 2012 summer REU interns, is here in the lab working on her Echinacea hybridization project. She spent the morning meticulously extracting achenes from the Echinacea pallida x angustifolia crosses she performed last summer.
By Marie Schaedel
Most Echinacea heads have achenes with a clear weight gap between full and empty, which is useful for lab purposes. Sometimes, however, there is no clear difference in weight for full and empty achenes. This winter, I took a closer look at achenes from Echinacea heads that had a continuous pattern of variation in weight to see if there was another way to distinguish between full and empty achenes when the weight test failed. To do this, I reweighed achenes from 20 samples that lacked an obvious weight cutoff. After plotting the new weights in R, I gathered X-ray images of the achenes closest to my best-guess cutoff weight for each sample. I found that more than half of the samples I tested had an intermediate transition weight that divided the full from empty populations of achenes. For ambiguous cases in which there is no clear difference in weight between full and empty achenes, this transition weight can be used to predict the cutoff value.
Read more:What to do when the Weight Test Fails.pdfAppendix
Figures

Figure 1

Figure 2

Figure 3

Figure 4

Figure 5
X-Ray Images

12139.1_top_red

14011.1_bot_bac

14142.1_bot_wht

14142.1_top_wht

14142.2_bot_grn

14142.2_top_grn

14143.1_bot_grn

14143.1_top_grn

14143.2_bot_wht

14963.2_bot_bac

14963.2_top_bac

17019.1_bot_wht

17019.1_top_wht

17134.2_bot_wht

17134.2_top_wht

17172.1_bot_red

17172.1_top_red

17294.1_bot_clr

17294.1_top_clr

6417.1_top_blu
Photograph Images

12139.1_top_red: Close-up of two achenes with a transition weight

14142.1_top_wht: Close-up of two achenes with a transition weight




















David Lowenstein, a PhD student from University of Illinois at Chicago, came to the lab at the Chicago Botanic Garden in December to use our x-ray machine for his research project. One of the species he using in his project is Echinacea purpurea.

David x-raying a sample of Echinacea fruits (achenes).
Here’s what David had to say about his project…
I am studying pollinators in Chicago and whether vegetable crops and wild flowers in various neighborhoods in Chicago receive sufficient pollination. To investigate this, I used a mobile garden consisting of eggplant, cucumber, and purple coneflower. Potted plants of these species were transported to Chicago neighborhoods and left outside for four and half hours. I observed bee visits to focal plants, sampled the neighborhood floral community, and allowed the plants to mature under pollinator exclusion netting in the UIC greenhouse to measure pollination services. I measured pollination of purple coneflower by X-raying the achenes at the Chicago Botanic Garden and recording the proportion of full achenes from each Echinacea flowerhead. I found a positive relation between bee abundance and human population density, and I will use data from X-rayed achenes to determine if pollination services are also related to population density or other land cover variables. To read more about this project that will continue in summer 2013, visit http://www.uic.edu/labs/minor/urban_pollination.html
Here are three of his x-ray images. If you click & enlarge these images, you will see that some achenes are empty and others have embryos.



Mindy Runge & Ale Mendoza participated in a mini-internship in the Echinacea lab at the Chicago Botanic Garden this fall. They are in Lynn Westley’s Plant Biology class at Lake Forest College.
Mindy & Ale investigated seed set in Echinacea heads from a prairie remnant in western Minnesota. They compared seed set in tops and bottoms of heads (florets at the bottom of a head flower first). In the process of answering their question, they learned how to dissect achenes from Echinacea seed heads, how to operate our high-tech seed balance, and how to organize datasets.

Ale and Mindy dissecting seedheads
This week we said goodbye to many of the students and volunteers who are traveling for the holiday season. Maria Wang left behind her dicanthelium to travel to visit with family in the UK. One of our volunteers, Bill W., is preparing for a trip to Panama. While a few people will be coming in next week, we won’t see most of our volunteers until next year. We’ve made a lot of progress in the past few months. Here is a brief summary of what we’ve accomplished since September:
1. New faces
We hosted several new interns this fall. Five students from Lakeforest College completed a four week internship on various projects (you can read about some of their work on the flog) . Marie (Carleton College) has been doing great work for an ongoing study on flowering phenology in prairie remnants. We will miss her when she goes back to school.
2. Lots and lots of counting
We have made a big dent in counting the achenes harvested in 2011 from the main experimental plot. We finished all counting for the 1999 experiment (4000s batch) and have begun counting for the rest of the 2011 harvest. And of course, no counting endeavor would be complete without a motivational whiteboard graphic.

3. New protocols
Because there are relatively few Echinacea heads to process from this year, we have an opportunity to try out different protocols and improve our methods. Our new protocol for selecting a random sample of seeds will improve our ability to assess pollination based on seed weight. We are also trying out new techniques for extracting seed heads and separating the dust.

That about sums it up. These details (protocols, cleaning, etc.) may seem minor, but they are important for creating reliable data.
|
|